 Therefore, when this light passes through a gas, the gas atoms may absorb certain wavelengths to produce a line absorption spectrum.īlack hydrogen absorption lines are at the same wavelength as the bright hydrogen emission lines. The energy levels in the atom have definite values. 1 2 Soon the term referred to a plot of light intensity or power as a function of frequency or wavelength, also known as a spectral. This is called a line absorption spectrum. Line spectra are produced when electrons move from one energy level within an atom to another energy level. In the physical sciences, the term spectrum was introduced first into optics by Isaac Newton in the 17th century, referring to the range of colors observed when white light was dispersed through a prism. So if a continuous spectrum travels through a cooler gas, it will ‘pull out’ or absorb certain wavelengths of light to produce a series of dark lines on a continuous spectrum of light. When a gas is cool, it absorbs the same wavelengths of light as it would emit when it is hot. A spectral line is a weaker or stronger region in an otherwise uniform and continuous spectrum, resulting from emission or absorption of light in a narrow. As the electrons move closer to or farther from the nucleus of an atom (or of an ion), energy in the form of light (or other radiation) is emitted or absorbed. These wavelengths are different for different elements and compounds. It’s important to know that only certain wavelengths of light are emitted. A gas cloud on its own, without a light source behind it, produces a line emission spectrum. Spectral lines are produced by transitions of electrons within atoms or ions. Definition: A line spectra consist of a number of well-defined lines each having a particular frequency or wavelength or color. Hot gases don’t produce a continuous emission spectrum.Ī hot gas only emits certain wavelengths of light to produce bright lines on a dark background. In a total consumption burner, both gases (fuel and oxidant) and the sample, come. There are two types of aspirator-burner used, total-consumption burner and premix chamber burner. When a gas is very hot, it doesn’t emit all wavelengths of light. The burners used as excitation sources in analytical flame spectroscopy should produce a stable and reproducible flame supplied with the analytical sample solution. A light source, such as a star or a filament bulb, gives a continuous emission spectrum. The continuous spectrum definition is all of the values in the spectrum without any gaps, skips, or breaks. This incredible rope is available in diameters ranging from to 1 ¼ and a tensile strength 2,900. 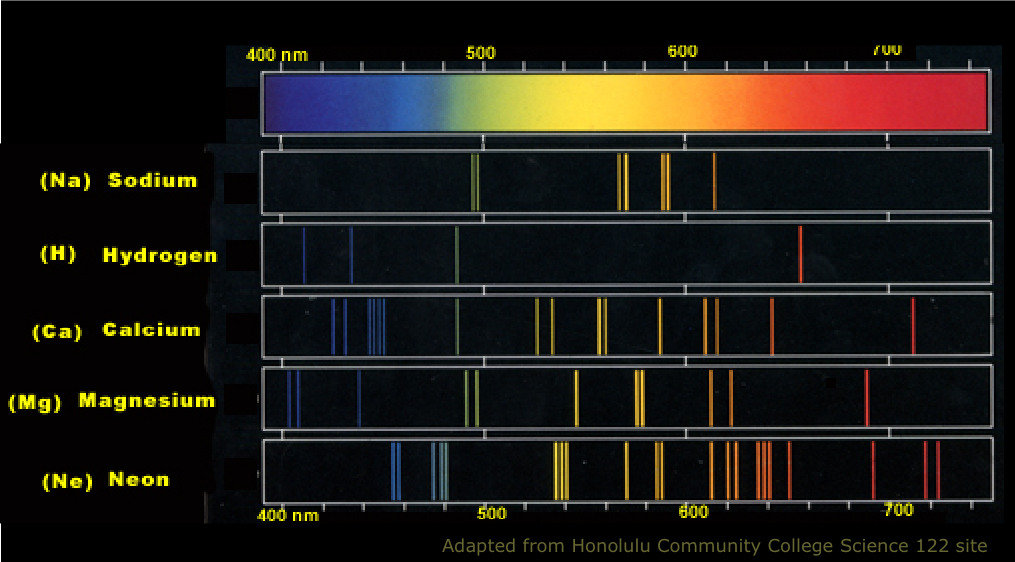 
The S-12 includes a special coating from Pelican Rope and offers extremely high stretch. It is known for its resistance to chemicals, abrasion, and flex fatigue. This is called a continuous emission spectrum. In general terms, a spectrum is the set of values between two extremes. This 12 strand braided Spectra Cord floats on water and resists corrosion. This light, when seen through a prism or diffraction grating, shows all wavelengths of visible light. 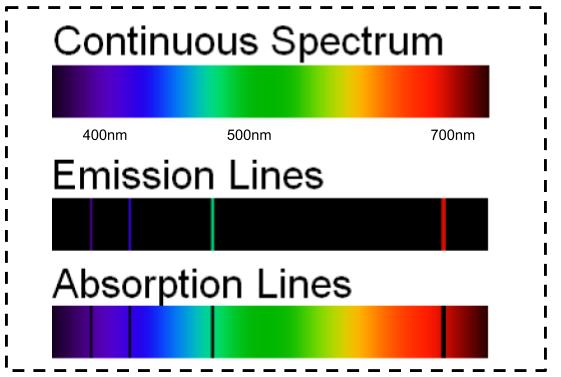
Studying the line spectra produced by hot gases and absorbed by cooler gases allows us to identify the elements in stars. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
